How Malaysia is setting itself up to provide biosimilars across the Islamic world as well as Asia.
Inno Bio Ventures, a Malaysian state-owned company originally set up to create drug security by ensuring that the country was self-sufficient in terms of medicine supply, has been making its first moves into the burgeoning biosimilars market through two significant joint ventures with a Korean and an Iranian firm.
Malaysia’s location in Southeast Asia combined with its Muslim-majority population means that the country is increasingly seen as a strategic partner for international companies looking to penetrate both ASEAN and Middle Eastern markets.
In this vein, Alteogen, a South Korean firm, this year signed a memorandum of understanding with Inno Bio Ventures to establish a joint venture for the development, clinical research, production and marketing of biosimilar drugs. “The idea is partly for us to utilize our connections to the Muslim world. The South Korean entity is seeking to deploy Malaysia as a hub to penetrate Middle Eastern markets,” elaborates Tan Sri Rahman Mamat, chairman of Inno Bio Ventures.
Meanwhile the company has received international recognition from the D-8 Organization for Economic Cooperation – a grouping of heavily populated but geographically disparate Islamic countries (Bangladesh, Egypt, Nigeria, Indonesia, Iran, Malaysia, Pakistan, and Turkey) – for its biosimilar product invention efforts. This project has been undertaken in collaboration with the Iranian outfit Ayrogen Pharmed.
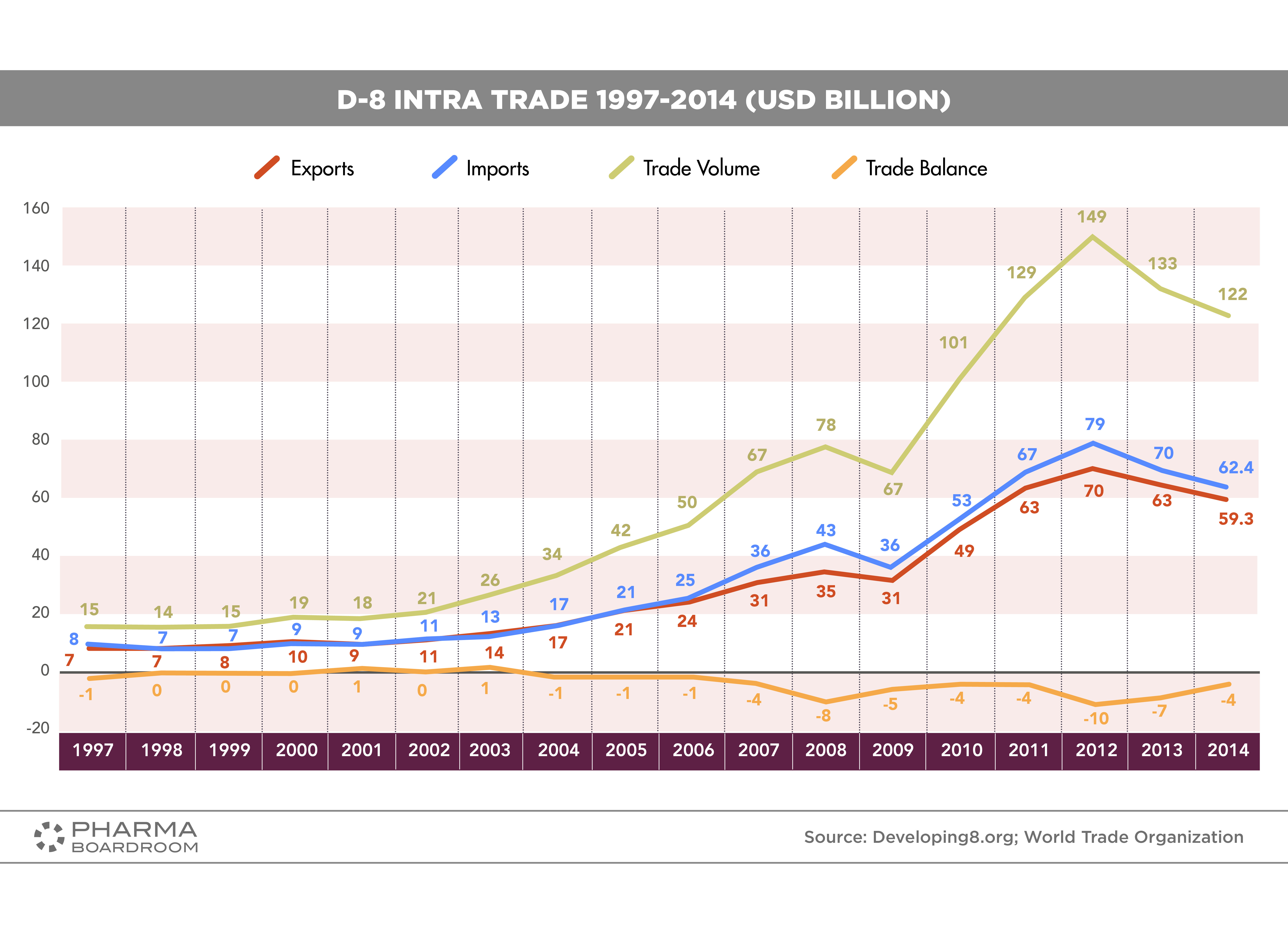
“This has been labelled the best joint venture effort to develop biosimilars as a solution for the D-8 countries,” explains Rahman Mamat. “Our project, when realized, aims to bring benefit not just to the Malaysian market, but also to patients across the Islamic world.” The potential combined market size of the D-8 countries is enormous, accounting for around one billion people, 60 percent of all Muslims, and close to 13 percent of the entire global population. Moreover, Intra-D8 trade stood at USD 122 billion in 2014 (see below graph).
 However, the geopolitical uncertainties of the Middle East have already hindered Inno’s ambitions to bring their Iranian joint venture products to the domestic Malaysian market. Rahman Mamat notes that, “we encountered a setback when sanctions were re-imposed on Iran. As a result, Ayrogen Pharmed’s products were only recognised in Iran, and not by the FDA or the EMEA. As this situation was unacceptable for the Malaysian authorities, it meant that we had us to delay our project in Malaysia.”
However, the geopolitical uncertainties of the Middle East have already hindered Inno’s ambitions to bring their Iranian joint venture products to the domestic Malaysian market. Rahman Mamat notes that, “we encountered a setback when sanctions were re-imposed on Iran. As a result, Ayrogen Pharmed’s products were only recognised in Iran, and not by the FDA or the EMEA. As this situation was unacceptable for the Malaysian authorities, it meant that we had us to delay our project in Malaysia.”
Conversely, the Iran sanctions have meant that Ayrogen is looking to lean on Inno, and Malaysia, a lot more, potentially fast-tracking the country’s transition into a biosimilars export hub. Rahman Mamat posits that, “There is a strong potential for exporting biosimilars from Malaysia, conditional on us transferring the technology. Given the political conflicts between Iran and the Arab world, Ayrogen’s intention is to use Malaysia as a hub as we are seen as the intermediary for its products to be accepted in the Arab states of the Middle East. We are fortunate as the Middle Eastern countries are connected to Asia through Islam.” Malaysia is also being seen as a gateway to the ASEAN region, with Inno already having been granted permission by Ayrogen to export products to China, for example.


